A Chemical Reaction Is At Equilibrium When The Rate Of Forward And Reverse Reaction Are – Chemical equilibrium is the state where both product and even reactants are present in concentration.
They have no further tendency to change with the time.
You must have read or learn about Chemical equilibrium in your school and college days. At times we do not understand what it is all about. But when you go deep down the subject then it becomes easier to know what is happeing.
However, the case where everything can be solved as well. The same is the case where you will get to know about Chemical equilibrium.
However, the case to solve when the rate of forward and reverse reactions are?
A Chemical Reaction Is At Equilibrium When The Rate Of Forward And Reverse Reaction Are
Talking about forward and reverse reactions both of these are produced when reactants goes from left to right in reversible reactions.
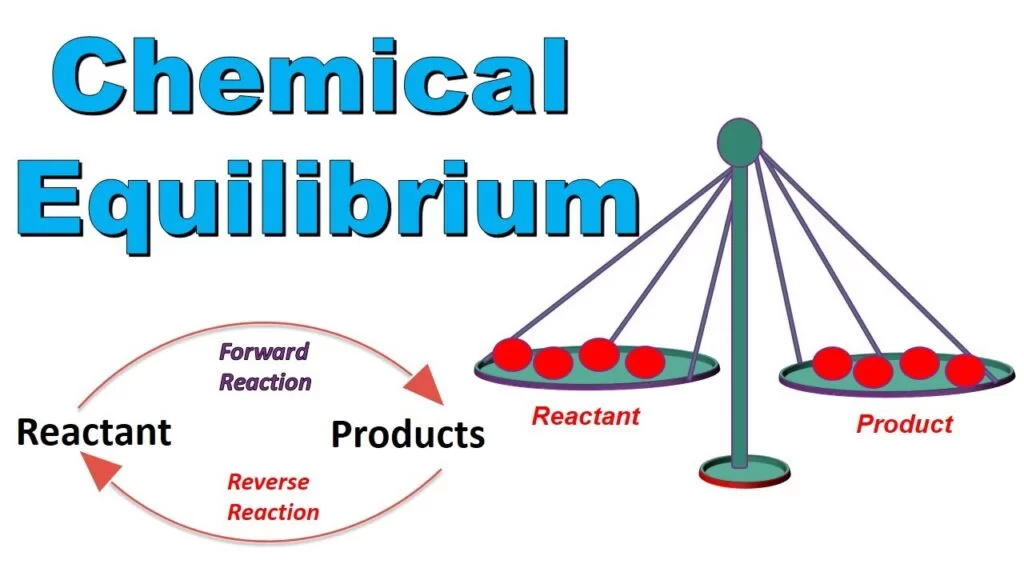
Whereas talking about the reversible reaction they are produced from products and goes from right to left.
The concept of Chemical equilibrium was came into 1803. It was found that there are some reactions that are reversible.
Then and there studies continued and also assisted many researchers to know about them. However the case where everything was settled with the passing time.
At constant temperature and pressure one must consider the gibbs free energy that stands for G.
but on the other hand at constant temperature and volume one must consider the Helmholtz free energy.
Hence there are different process that needs to be undertake to help people understand.
However, it might take sometime for you to understand. But never the less you can easily help yourself.
Get to know about Chemical equilibrium
With the help of web everything has been simplified. No matter what is your requirement you can easily withstand with every result.
In this manner Chemical equilibrium is one of those state.
There are two stages forward and reversible reactions. Both of these vary from each other in some context.
Hence in this manner you have to understand with which force do they work.
Moreover once the concept is clear then you can easily determine Chemical equilibrium.
- The Forgotten Love Novel Julia And Robert Read Online PDF
- Feedce.com+Download Delete Video Recover
- Navneet 21 Set for SSC 2022 PDF Download
- Love like The Galaxy Novel English Translation Read Online